editorial platform
Editorial Platform for EBM Guidelines
The platform of EBM Guidelines has been developed by Iscientia, and gives health care professionals access to different databases of EBM Guidelines. Check our page on EBMG for more information!
Besides this global information, every country can add specific national guidelines from different guideline developers, or can add contextual notes to international guidelines.
In order to do so, iscientia has developed an editorial platform:
- to review, adapt and update international or national guidelines,
- to write and implement national guidelines,
- which is user friendly and enhances collaboration between editors, proofreaders and authors,
- newly or modified information can be immediately published after validation
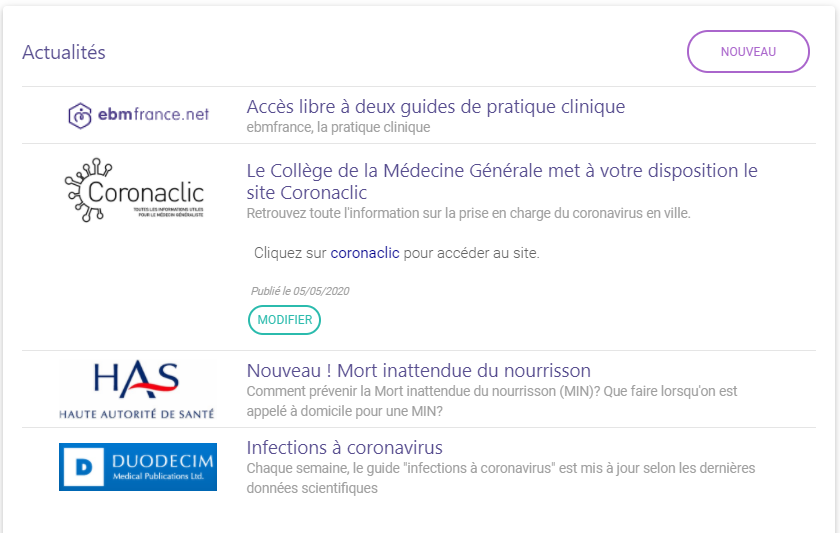
News and newsletters
It is possible to publish news items on a daily basis, just create and publish the news item in a few clicks, add an illustration and external links.
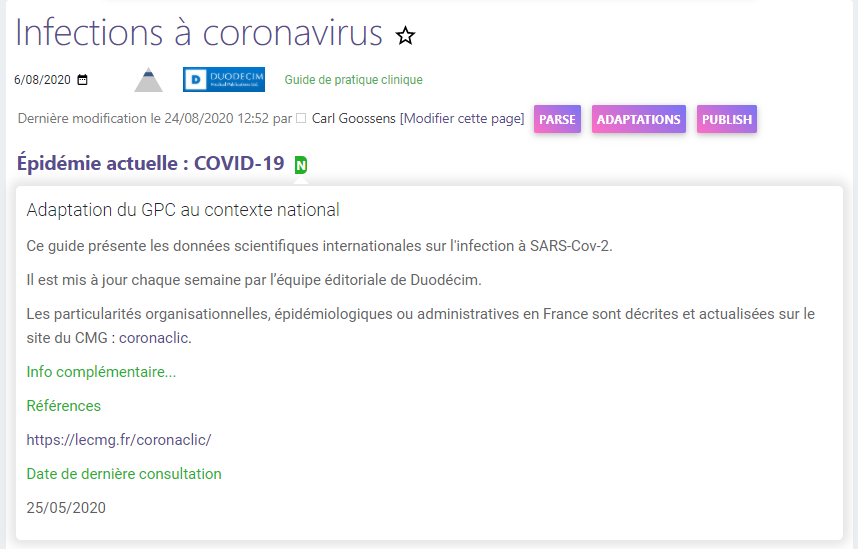
Contextual information
It is possible to add, edit and publish additional contextual information, such as specific national information, with illustrations, tables and links to external websites.
This information is integrated in an editorial platform, and after peer review and validation by the chief editor, it can immediately be published on the public site.
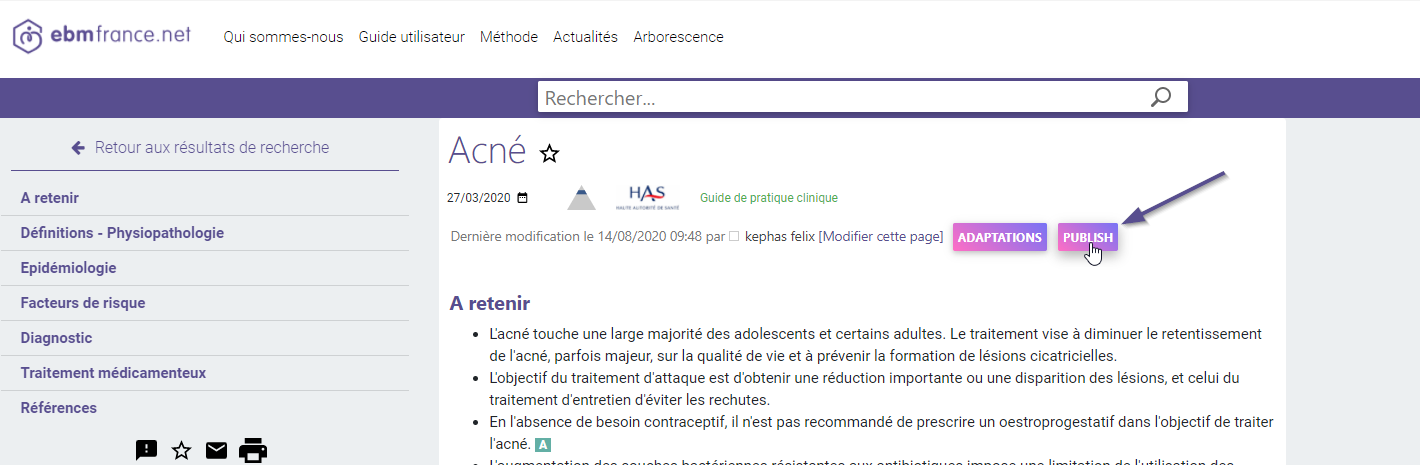
Create and publish new national guidelines, evidence summaries, tools and more
The editorial platform supports the possibility to create a new guideline, add illustrations, tables and links, and get them published as soon as they have been finalized.
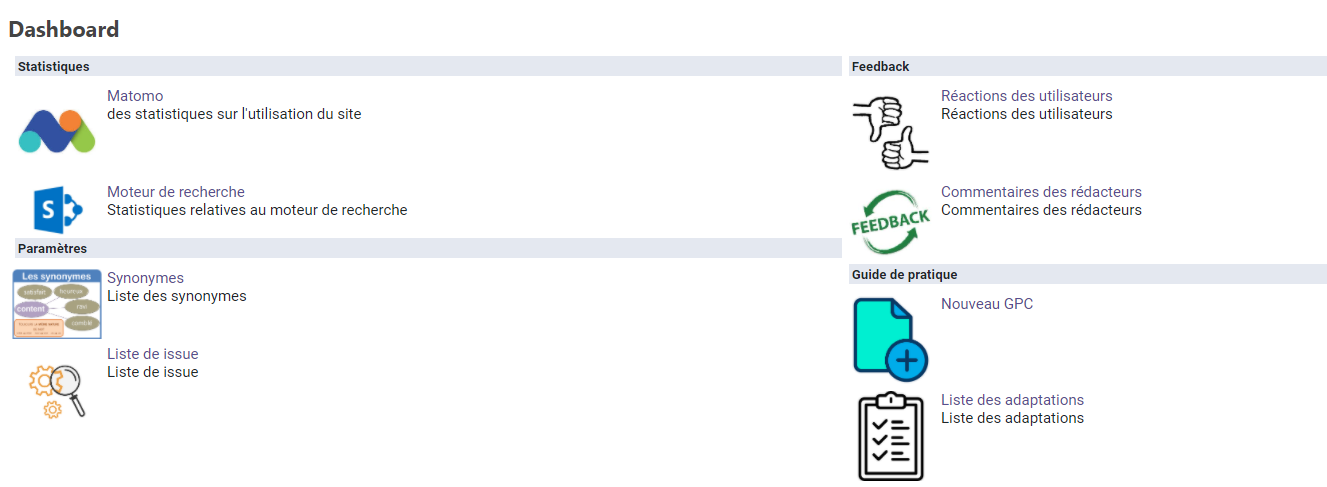
Editorial Dashboard
The medical authors, editors and proofreaders have access to a dashboard, in order to
- create new guidelines,
- view statistics,
- view feedback from external users,
- log issues regarding the site,
- add synonyms,
- validate adaptations